
Have been multiplied in a way that the function is normalized. The coefficients within the hybrid wave function This combination is called hybrid orbital and the respective approach hybridization. This expression is a linear combination of two orbitals, one from lithium is a combination of Φ 2s and Φ 2p. In the following form, the equation allows for this. Nevertheless, there is still an opportunity to cling to the simple principle where molecular orbitals emerge from an overlap between two orbitals and that both sides introduce just one orbital. The solution above has been obtained exactly in this way. Thus, we shall combine all atomic orbitals of a certain energy level and solve the respective secular equation. 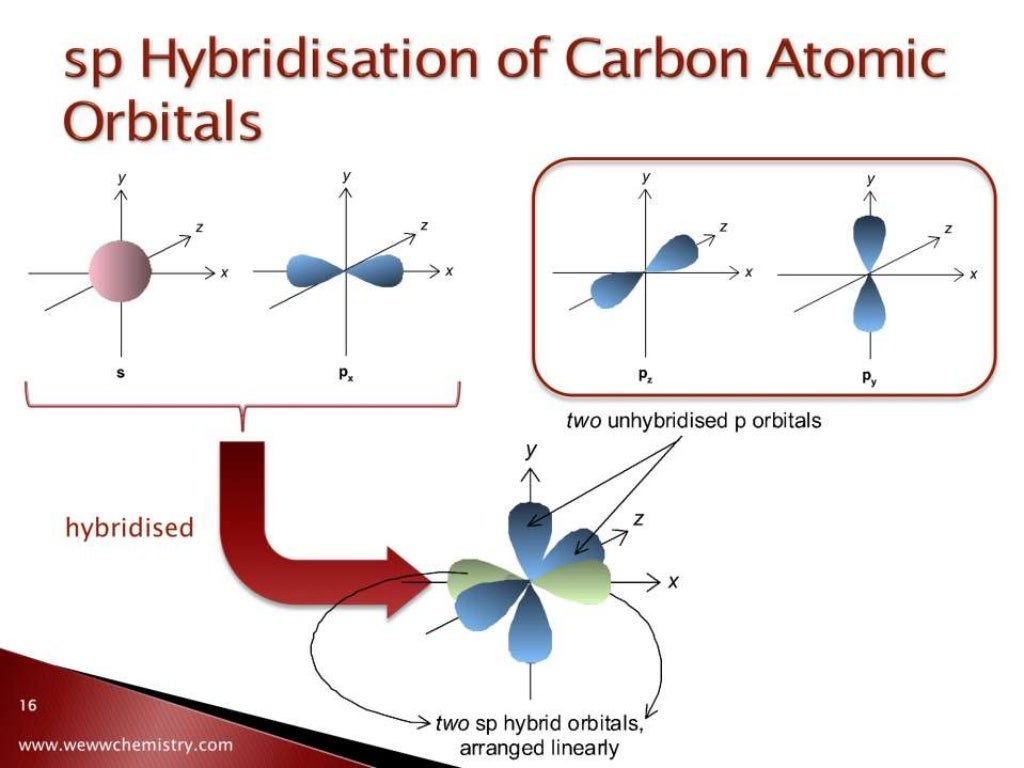
The 2p-AO of lithium needs to be introduced because the 2p-AO is only slightly higher in energy and the condition of similar energies for combining AO is met in the same way as it is with the 2s-AO. Thus, in lithium hydride we find an inner shell (1σ)² that is almost identical with the respective atomic orbital, and a bonding 2σ-MO occupied by two electrons. the Li atom with 1s-, 2s- and 2p-atomic orbitals and Φ 1s, Φ 2s and Φ 2p as wave function and a hydrogen wave function Φ H) produce (neglecting a minor involvement of Φ 1s) according to Ransil (1960) the following molecular orbitals Ab initio calculations (derived from the self consistent field approach) with a minimal basis (i.e. To make four bonds, carbon would have to “decouple” its s-electrons onto the p shell.Now we want to consider the example of Lithiumhydride LiH and apply the LCAO description of molecular orbitals. This would only allow carbon to make 2 bonds since it only has 2 unpaired electrons. If we look at the carbon atom atomic orbitals, we’ll see the 2 electrons on the 2s and 2 electrons on the 2p shells. □ Each bond takes 2 electrons to complete. Ok, now when we know that hybridization is a model and not an actual process, let’s look at how this “process” happens. Hybridization is a mathematical model that describes how the atomic orbitals would’ve looked like based on the observable molecular orbitals. The atomic orbitals don’t actually change before going into the bonding with other atoms. There’s no actual “process” that happens to orbitals that causes the hybridization. Let me emphasize one more time that hybridization is a mathematical model. Not a complete s- or a p-orbital, but rather something with a partial s- and partial p-character. 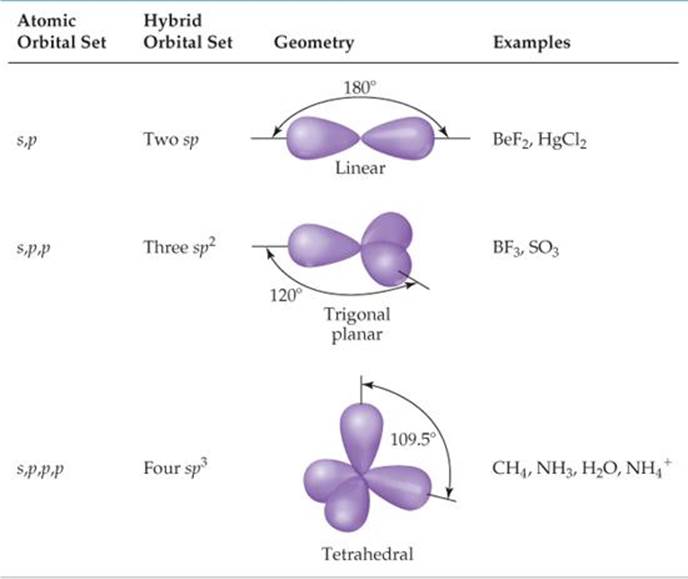 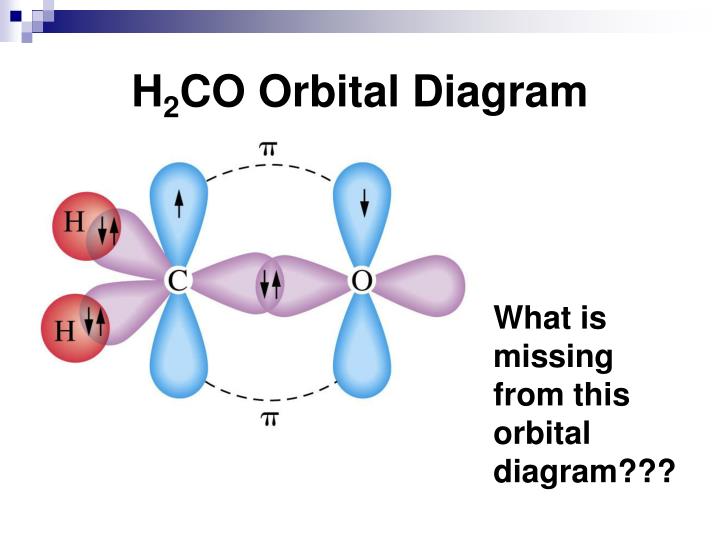
Pauling proposed sort of a combination of the orbitals giving you an orbital that has partial characters. In 1931 the twice Nobel Laureate Linus Pauling proposed the model of “mixing” the orbitals or “hybridizing” them to account for the observed bonding pattern. So, how can we have 109.5° bond angles made by the orbitals which are at 90° to each other? □□♂️□ We also know that VSEPR describes the 3D shape of the second period elements reasonably well.Īnd here we have a problem: the atomic orbitals are at 90° to each other, while the VSEPR theory predicts the 3D structure of, say, methane (CH 4) to be tetrahedral with bond angles around 109.5°. We all know from general chemistry that the s-orbital is spherical, and p-orbitals are dumbbell-looking orbitals oriented along the x, y, and z axes of the Cartesian system.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
